Foundayo Weight Loss Pill: A Practical Guide to the New Oral GLP-1 Treatment
A New Phase in Weight Management
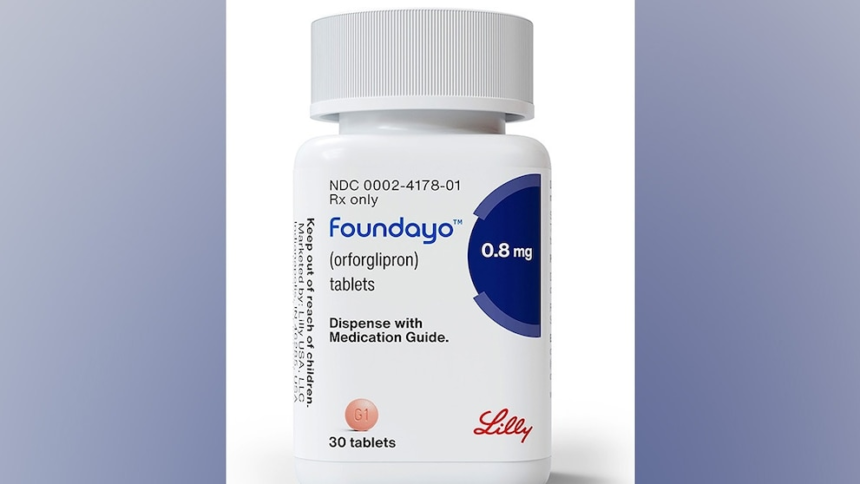
The approval of Foundayo (orforglipron) marks a notable shift in how weight loss and obesity are being treated globally. For years, the most effective medications in this space—particularly GLP-1 receptor agonists—have largely been injectable. That model, while clinically effective, has also created barriers related to convenience, cost, and patient acceptance.
- A New Phase in Weight Management
- What Foundayo Is and How It Works
- Why the Shift From Injections Matters
- Clinical Results: What the Data Shows
- Real-World Experience: What Patients Report
- Dosage Structure and Treatment Approach
- Side Effects and Safety Considerations
- Cost, Access, and Insurance Dynamics
- How Foundayo Compares to Alternatives
- The Broader Impact on Healthcare
- What Comes Next for Weight Loss Treatments
- Conclusion: A Shift Toward Practical, Everyday Treatment
Now, with the introduction of Foundayo, a once-daily oral pill, weight management is entering a more accessible phase—one that aligns more closely with everyday routines rather than clinical interventions.
Regulators in both the United States and the United Arab Emirates have moved quickly to approve the drug, signaling the urgency of addressing obesity as a major public health issue. The U.S. Food and Drug Administration approved the medication in just 50 days, one of the fastest timelines for a new molecular entity in recent years.

What Foundayo Is and How It Works
Foundayo belongs to a class of medications known as GLP-1 receptor agonists. These drugs mimic a naturally occurring hormone—glucagon-like peptide-1—that regulates appetite and blood sugar.
The mechanism is straightforward but powerful:
- It slows digestion
- It increases feelings of fullness
- It reduces hunger signals
- It helps regulate blood glucose levels
Unlike earlier GLP-1 drugs, Foundayo is taken as a daily oral tablet, eliminating the need for injections or strict timing protocols.
A key advantage is flexibility: the pill can be taken with or without food, at any time of day. This distinguishes it from other oral alternatives, such as semaglutide tablets, which require fasting conditions before consumption.
Why the Shift From Injections Matters
The transition from injections to pills addresses a critical issue in obesity treatment: low adoption rates.
Despite the proven effectiveness of GLP-1 medications, fewer than one in ten eligible patients currently use them. Several factors explain this gap:
- Fear or discomfort with injections
- Lifestyle inconvenience
- Storage requirements (some injections require refrigeration)
- Social stigma around obesity treatment
Foundayo directly targets these barriers. By simplifying administration, it makes long-term treatment more feasible for a broader population.
Medical professionals have emphasized this benefit. As one expert noted, “People are used to taking pills. Getting a shot throughout your life is not fun.”
Clinical Results: What the Data Shows
Clinical trial data has been central to Foundayo’s rapid approval.
In trials lasting up to 72 weeks, patients who remained on the highest dose of the medication experienced:
- An average weight loss of 27 pounds (approximately 12.4% of body weight)
- Significant improvements in cardiovascular risk markers, including:
- Cholesterol levels
- Waist circumference
- Blood pressure
Another dataset reported weight loss ranges between 8 to 25 pounds, depending on dosage and adherence.
These outcomes position Foundayo as a long-term weight management tool, not a short-term solution.
Real-World Experience: What Patients Report
Early patient experiences provide insight into how the drug performs outside controlled trials.
One participant in a clinical study described a noticeable shift in appetite within days:
“I had about a portion, and then realized I was perfectly satisfied. I didn’t need any more food.”
The same patient reported losing 25 pounds over 18 months, with sustained results even after discontinuing the medication.
The most commonly reported effect was slower digestion, which contributed to earlier satiety.
Dosage Structure and Treatment Approach
The FDA outlines a gradual dose escalation strategy for Foundayo:
- Start at 0.8 mg
- Increase to 2.5 mg after 30 days
- Then to 5.5 mg, with further increases possible up to 17.2 mg
This incremental approach is designed to improve tolerability and reduce side effects.
Importantly, Foundayo is not intended as a standalone solution. It is prescribed alongside:
- A reduced-calorie diet
- Increased physical activity
Side Effects and Safety Considerations
Like all GLP-1 medications, Foundayo comes with potential side effects.
Common reactions include:
- Nausea (reported in 29–34% of patients)
- Constipation (22–25%)
- Diarrhea (21–23%)
- Vomiting (13–24%)
- Abdominal pain and headache
More serious risks include:
- Pancreatitis
- Kidney complications
- Low blood sugar
- Gallbladder disease
The drug also carries a boxed warning for thyroid C-cell tumors, meaning it is not suitable for individuals with certain thyroid conditions.
Cost, Access, and Insurance Dynamics
Affordability is expected to play a decisive role in adoption.
In the United States:
- Patients with commercial insurance may pay as little as $25 per month
- Self-paying individuals may face costs starting at $149 per month, increasing with dosage
For Medicare Part D beneficiaries, pricing could begin around $50 per month, depending on eligibility.
In the UAE, pricing has not yet been officially announced, but similar benchmarks are likely to apply.
Access will depend heavily on:
- Insurance coverage policies
- Whether obesity treatment is classified as essential care
- Long-term affordability, since treatment is ongoing rather than short-term
How Foundayo Compares to Alternatives
Foundayo enters a competitive and rapidly evolving market.
Other GLP-1 options include:
- Injectable drugs like Wegovy and Ozempic
- Oral semaglutide (currently under regulatory review in some regions)
Preliminary data from the ORION study suggests that oral semaglutide may deliver slightly greater weight loss and lower discontinuation rates. However, these findings are based on indirect comparisons rather than direct head-to-head trials.
Patient preference remains a critical factor. Some individuals prioritize convenience, while others may opt for treatments with longer clinical track records.
The Broader Impact on Healthcare
The introduction of Foundayo reflects a broader shift in how healthcare systems approach obesity.
Obesity is linked to more than 200 associated conditions, including:
- Type 2 diabetes
- Hypertension
- Cardiovascular disease
- Certain cancers
Expanding treatment options—especially those that are easier to integrate into daily life—could reduce long-term healthcare burdens.
The UAE’s decision to fast-track approval highlights a strategic focus on early access to innovative therapies, positioning the country among the first to adopt next-generation obesity treatments.
What Comes Next for Weight Loss Treatments
The next phase in this space will likely focus on differentiation rather than availability.
Key factors shaping future competition include:
- Comparative effectiveness
- Side effect profiles
- Cost structures
- Patient adherence rates
As more oral GLP-1 options enter the market, treatment decisions will become increasingly personalized.
Conclusion: A Shift Toward Practical, Everyday Treatment
Foundayo represents more than just another weight loss medication—it signals a structural change in treatment delivery.
By removing many of the practical barriers associated with injections, it brings obesity care closer to everyday life. However, questions around cost, insurance coverage, and long-term effectiveness remain central to its broader impact.
For patients, the decision will ultimately come down to a balance of convenience, affordability, and clinical outcomes. For healthcare systems, it marks a step toward more scalable and accessible obesity management strategies.